市场情报
-
2024.04.15
2024医疗器械展会提醒:又一项新指导原则发布,助力哪些医疗器械细分领域崛起?
之前发布的《重磅!又一项新注册审查指导原则发布,创新医疗器械审评高质量发展再提速》和《速览|浅析医疗器械可用性工程的合规性》两篇文章中,我们介绍了可用性工程的基本概念、中外医疗器械可用性工程政策,并重点梳理了国家药监局器审中心3月19日发布的《医疗器械可用性工程注册审查指导原则》及其使用说明的内容要点。
阅读更多 -
2024.04.12
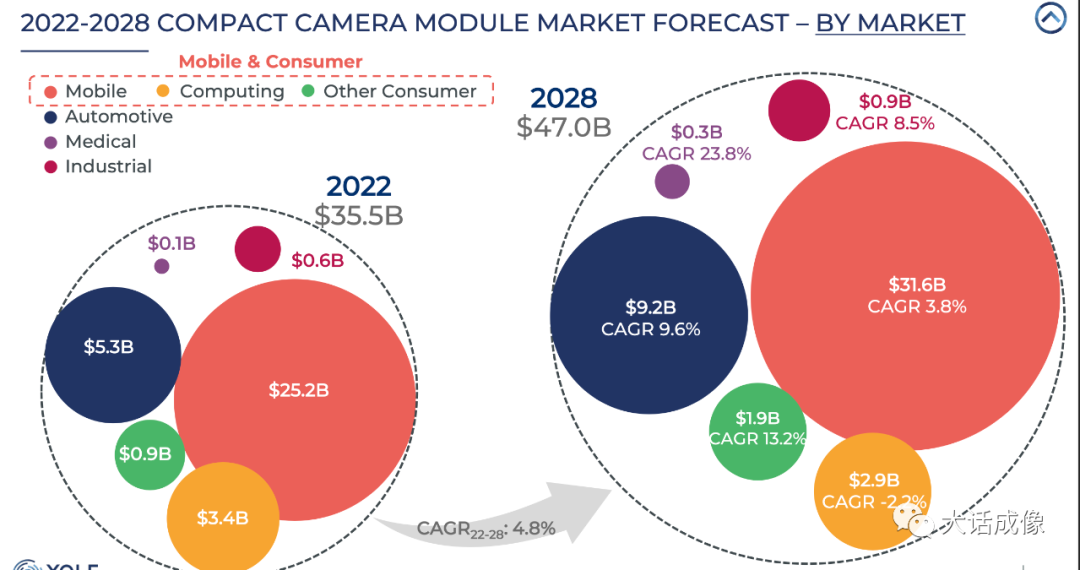
2024医疗器械展览会市场趋势调研之2023-2028年摄像头模组市场
本期的内容主要基于Yole发布的重磅报告《摄像头模块产业现状 2023》。接下来,我们将通过问答的形式,深入探讨摄像头模组市场的最新趋势和技术革新。
阅读更多 -
2024.04.10
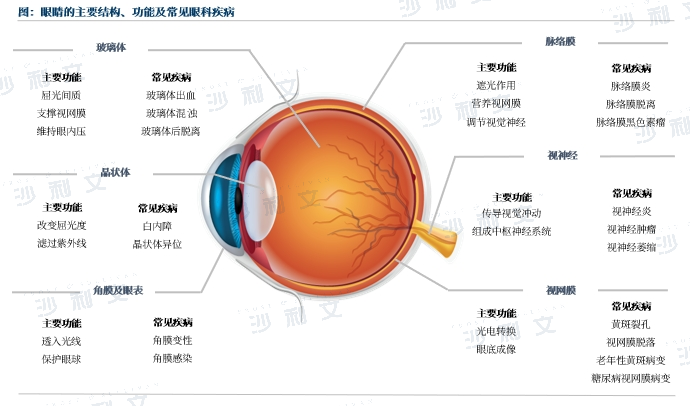
国际医疗器械展览会盘点沙利文发布《高端眼科医疗设备行业现状与未来发展趋势蓝皮书》
报告旨在剖析高端眼科医疗设备行业现状,全面展现行业技术发展、应用领域、上市产品情况,挖掘行业发展巨大潜力,分析市场发展背后的驱动因素以及未来发展趋势,以期为高端眼科医疗设备行业的参与者和关注者提供参考。
阅读更多 -
2024.04.08
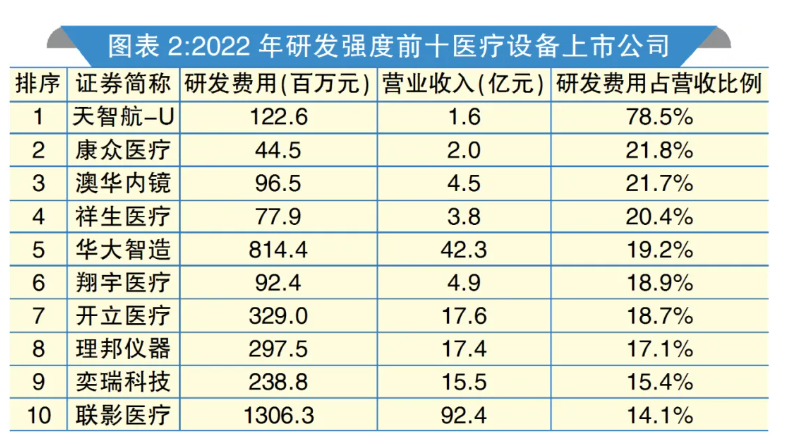
2024有源医疗器械创新论坛分享医械头部企业研发强度解析文章
推动发展的第一动力是创新,而创新离不开强有力的研发投入。创新领域斩获颇丰,与医疗器械上市公司在研发领域真金白银的投入密不可分。医疗器械头部力量高技术、高增长、高投入、高回报的特征已经凸显。
阅读更多 -
2024.03.29
2024医疗器械展览会听2024年两会上的医疗器械声音
本文速递了两会上的医疗器械声音,包括植入性医疗器械创新和规范管理,建议优化罕见医疗器械审批流程,科学突破高值一次性医疗器械使用限制等。点击立刻了解
阅读更多 -
上海医疗器械博览会看2024技术趋势有哪些?包括AI,综合医疗保健等
展望2024年,AI还可以将类似的音频体验带入手术室,用声音指导外科医生何时查看哪些屏幕,同时捕捉可以自动转录为笔记的手术背景,实现高度个性化的术后护理。

阅读更多 -
2024.03.20
2024上海高端医疗设备展Medtec China探发改委官宣:支持这些医疗器械
2024年3月10日,中国政府网公布了《产业结构调整指导目录(2024年本)》,公布了多项医械产业鼓励类目。新版目录已于2023年12月1日第6次委务会议审议通过,自2024年2月1日起施行,《产业结构调整指导目录(2019年本)》同时废止。
阅读更多 -
2024.03.19
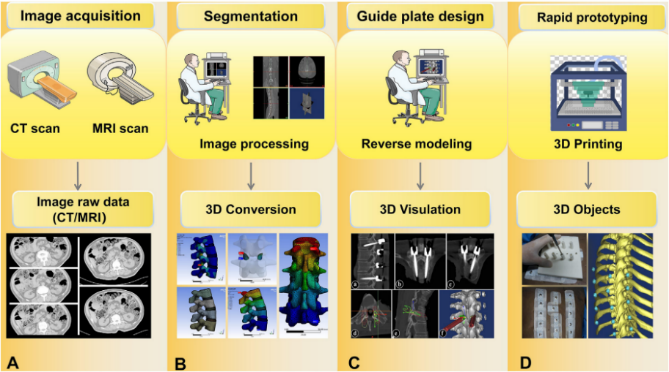
国际医疗器械展览会Medtec China观2023骨科创新转化新进展
创新推动发展,转化服务未来。推动高质量发展,关键是要依靠科技创新转化为发展动力。骨科创新转化不仅关乎技术的进步,更是关系长期受骨科疾病困扰患者生活质量的改善。新理念的萌生、新技术的突破以及新疗法的创新转化,都在不断推动着骨科领域的发展。
阅读更多 -
上海医疗器械展会Medtec China探奚廷斐教授专访:与中国镁合金产业筑梦同行
奚廷斐教授表示,坚持源头创新不易,稳住心态很重要,以团队进行医用镁合金转化的经验为例,产业化过程中遇到了诸多难点,如技术难点、经费难点、临床应用难点等。如今,他依然带领着团队奋战在生物镁合金产业转化一线上。
阅读更多 -
2024.03.18
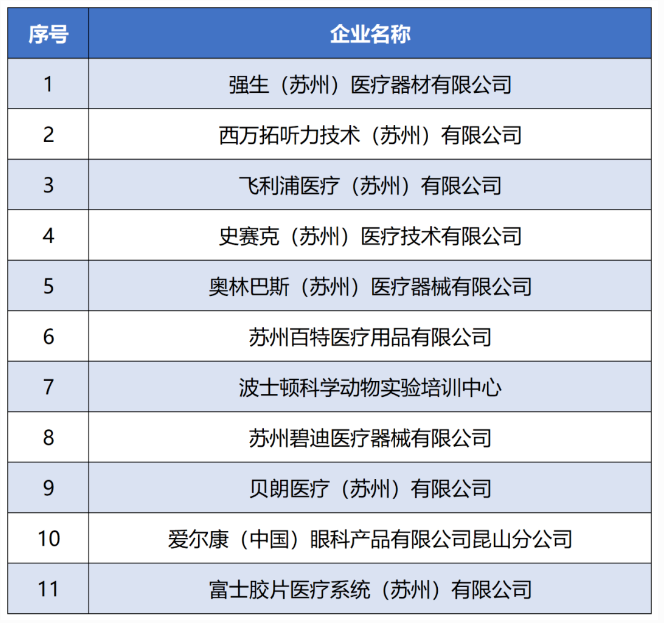
2024上海医疗设备展Medtec China探高端医械“要塞之地”——湖畔之城 苏州
世界顶尖工业设计奖项、拥有设计界“奥斯卡”之称的德国iF设计奖(iF DESIGN AWARD)2024获奖名单正式揭晓,苏州高新区企业——心擎医疗(苏州)股份有限公司的体外磁悬浮人工心脏MoyoAssist®从72个国家超10000件作品中脱颖而出斩获2024iF设计奖。
阅读更多










